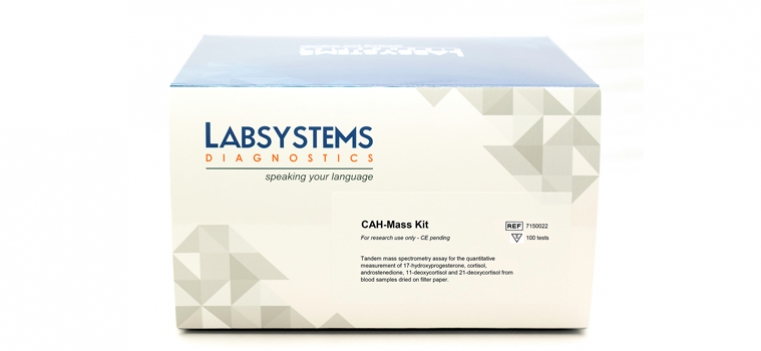
Quantitative determination of17-hydroxyprogesterone, Androstenedione, Cortisol, 11-deoxycortisol and 21-deoxycortisol in Dried Blood Spot Samples
Liquid Chromatography - Tandem mass spectrometry (LC-MS/MS) techniques allows for the simultaneous specific determination of 17-OHP and other steroids related with CAH such as androstenedione, cortisol, 11-deoxycortisol, and 21-deoxycortisol. Application of this technology to the determination of steroids in newborn blood spots significantly enhances the correct identification of patients with CAH and reduces the number of false-positive screening results when implemented as a second-tier analysis performed prior to reporting of initial newborn screen results.